What if Mandela really did die in prison. We were sold a narrative- again. Same script, with slight variations again and again. The actor playing the demented "Biden", Epstein's corps, etc.
Sunday, 29 March 2026
Saturday, 14 March 2026
Progesterone Misconceptions: A Few Things to Know
Leigh Erin Connealy
March 09, 2026
Progesterone is one of the most important hormones in the human body, yet it is also one of the most misunderstood. For decades it has been overshadowed by confusion around synthetic progestins, misconceptions about hormone therapy, and a tendency to reduce hormones to overly simple narratives. As a result, many physicians rarely consider progesterone outside of fertility or pregnancy, despite its broad effects on the brain, metabolism, inflammation, and tissue health.
Progesterone is not a rare or experimental compound. It is an inexpensive hormone the body naturally produces and that has been studied for decades. Yet persistent misconceptions about its role have prevented it from being widely used or properly understood.
To clear up some of this confusion, I want to break down a few of the most common misconceptions about progesterone:⬇️
1. “Estrogen is the ‘female’ hormone, not progesterone.”
Estrogen often takes center stage, especially in menopause care. Many women are told they can’t take estrogen without also taking progesterone, not because it’s equally important, but because progesterone helps protect the body from estrogen’s unopposed effects. Progesterone is essentially treated as a buffer.
Estrogen has been widely considered the primary female sex hormone, largely because of its role in stimulating tissue growth. Estrogen drives many of the physical features we associate with femininity, including breast development and the widening of hips during puberty. It’s essential for ovulation, menstruation, and pregnancy, and without it, reproduction would be impossible.
But progesterone may be even more important, and it rarely gets the attention it deserves. Its very name comes from its central role in fertility: “pro-gestation,” the hormone that supports pregnancy. But its influence extends far beyond reproduction. Progesterone is a master regulator. It stabilizes tissues, protects against inflammation, supports metabolic balance, and helps the body respond to stress in healthy ways. Unlike estrogen, which tends to stimulate, progesterone calms, shields, and repairs.
Interestingly, our bodies naturally produce only tiny amounts of estrogen—just fractions of a nanogram in the blood. In contrast, progesterone is produced in much larger quantities, especially after ovulation and during pregnancy. This is a reflection of progesterone’s broader importance.
On lab tests, estrogen and progesterone are measured in different units because they exist at vastly different concentrations in the body. For example:
Estrogen is typically measured in picograms per milliliter (pg/mL). A picogram is one-trillionth of a gram. It’s measured in such small units because it’s incredibly potent. The body only needs very small amounts of estrogen to have strong biological effects.
In contrast, progesterone is measured in nanograms per milliliter (ng/mL). A nanogram is one-billionth of a gram. This is because it’s produced in much larger quantities. In fact, one nanogram equals 1,000 picograms, so even if the number looks smaller, the actual amount of hormone is significantly greater.

During the luteal phase, a woman’s body typically produces 10 to 30 nanograms of progesterone per milliliter of blood, while estrogen (specifically estradiol) stays around 100 to 300 picograms per milliliter.
Since one nanogram equals 1,000 picograms, that means progesterone levels are roughly 100 to 300 times higher than estrogen.
This is like a teaspoon of estrogen compared to a gallon of progesterone.
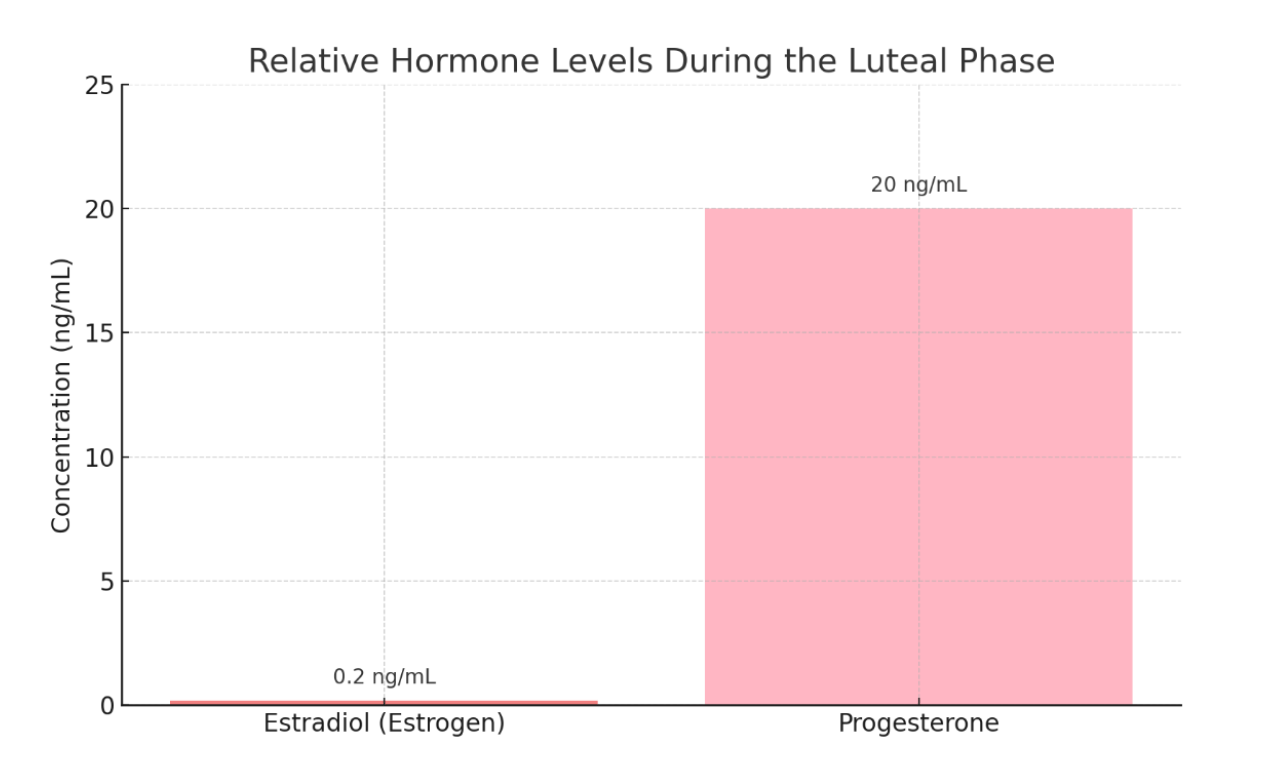

Even throughout most of the follicular phase, when progesterone levels are considered “low,” the concentration of progesterone remains elevated above estrogen.
It is only in the final day or two before ovulation that estrogen briefly surpasses progesterone in concentration. This short window marks the estrogen surge that triggers the LH spike and leads to ovulation, after which progesterone rises sharply and once again becomes the dominant hormone.

Most people often compare the two hormones by their numbers alone, without considering the units. This leads to a skewed perception—where estrogen appears “higher” or more dominant, simply because its levels are presented in smaller units. In reality, progesterone is often tens or even hundreds of times more concentrated.
Even during the third trimester of pregnancy, both estrogen and progesterone reach the highest concentrations they will ever attain in the human body:
Estradiol (the main form of estrogen) can rise to around 25,000–40,000 pg/mL.
But progesterone can reach approximately 200,000 pg/mL.
So, progesterone is still 5-8 times higher than estrogen.
When it comes to HRT, hormone balance, etc., estrogen usually gets most of the attention but our physiology shows us that progesterone is even more important. We need both hormones, but we need them in the right proportions.
Progesterone stabilizes tissues. It ensures that cells differentiate, and that tissues stay organized, functional, and safe.
Progesterone promotes a healthy cellular metabolism, helps cells use oxygen efficiently, reduces inflammation, balances blood sugar, and protects against overstimulation by estrogen. It calms the nervous system, supports deep sleep, stabilizes mood, and nurtures the uterine lining for pregnancy. It strengthens bones, protects brain tissue, and even supports thyroid function.
Estrogen is a growth promoter—it tells tissues to multiply and expand. But unregulated growth can quickly become dangerous if not balanced. And in general, we need little tissue growth (except during puberty, menstruation, pregnancy, and repair from injury).
Estrogen also stimulates excitatory neurotransmitters, it can raise cortisol, influence blood flow and cardiovascular health, cause weight gain, and increase fluid retention. In high amounts, it can heighten sensitivity to stress, amplify inflammation, and trigger histamine reactions. It affects nearly every system in the body—the brain, the bones, the immune system—sometimes in helpful ways, but often in ways that require careful regulation.
Without enough progesterone to balance it, estrogen’s effects can become excessive, and even dangerous. So, progesterone deserves more than a supporting role.
2. “Progesterone and synthetic progestins are the same.”
A common misconception when it comes to hormone replacement therapy (HRT) is that progestins are the same as progesterone. Progestins are often used interchangeably with progesterone, even in medical articles and mainstream media.
So what is the difference between bioidentical progesterone and synthetic progestins?
Micronized progesterone is a bioidentical hormone with a molecular structure identical to the endogenous progesterone produced by the ovary. Synthetic progestins have a completely different chemical structure from progesterone. They are made from various steroid “base” molecules, such as estrogen and testosterone.

Progestins get their name because they’re designed to bind to the progesterone receptor, anything that does can technically be called a “progestin.” But binding alone doesn’t mean they act like real progesterone. In reality, synthetic progestins often trigger very different, and sometimes opposite, effects in the body
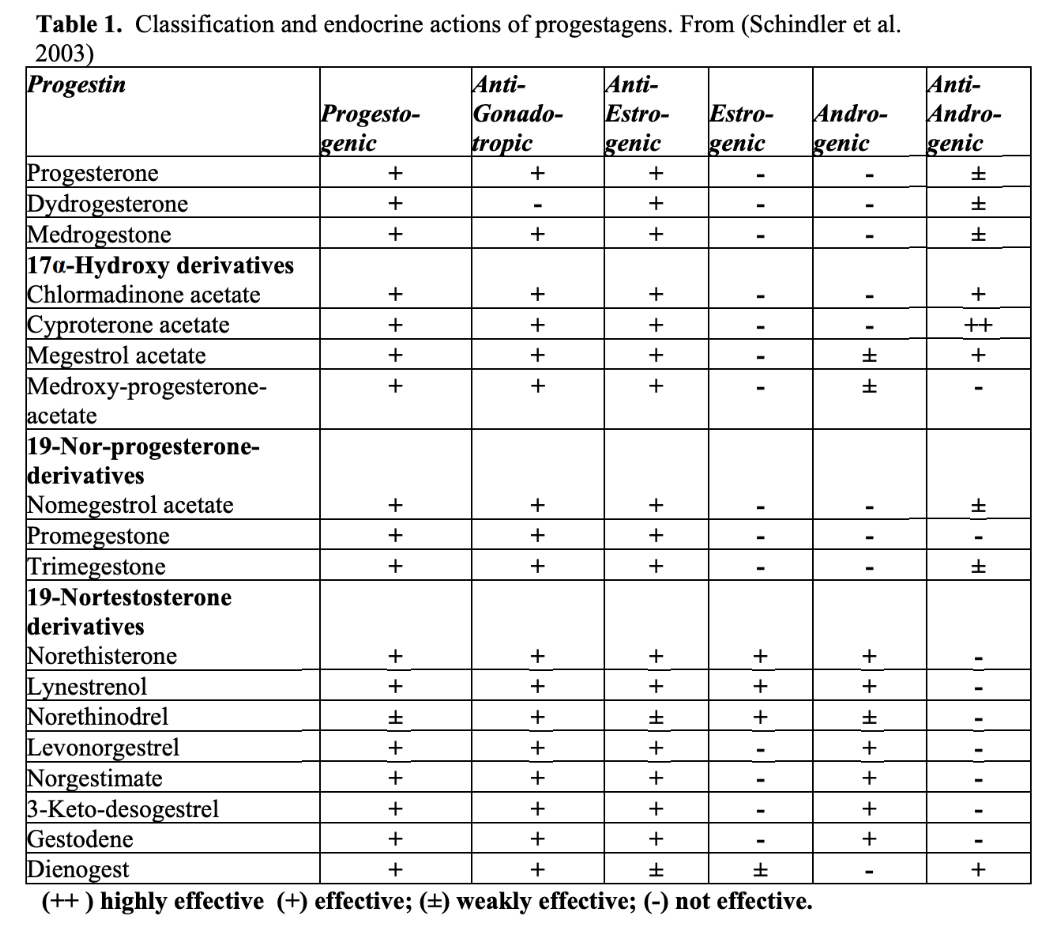
Incidentally, progestins often act more like estrogen, amplifying the harmful effects of estrogen and increasing the risk of cancer.
Conflating progestin with progesterone has led to widespread misunderstanding. This confusion has unfairly maligned bioidentical progesterone, which plays a protective role, opposes estrogen, supports healthy cell differentiation, and reduces the risk of hormonally driven cancers.

A few things to know:
One family of progestins, norethisterone, norethisterone acetate and norethindrone, in particular, are metabolized and actually become estrogens in the body (PMID: 12359276).
Medroxyprogesterone, the progestin most commonly used in HRT, acts like prednisone in the breast tissue and causes breast cells to proliferate. It increases the growth-promoting effects of estrogen, worsens insulin resistance, raises levels of insulin-like growth factor 1 (IGF-1), which is strongly linked to cancer development, and lowers sex hormone-binding globulin (SHBG), a protein that helps protect against hormone-driven cancers (PMID: 21336598).
Beyond cancer, progestins can cause cardiovascular damage and increase the risk of heart attacks, while progesterone has no adverse effects on the heart (PMID: 11108872).
Progesterone is “pro-gestation” and pro-fertility, hence the name, while progestins are used in contraceptive pills because of their anti-fertility effects.
Studies show that adding synthetic progestins to estrogen in hormone replacement therapy increases risk of breast cancer even when compared to the risk of using estrogen alone.
The famous Women’s Health Initiative trial of combined estrogen plus progestin had to be stopped early when overall health risks, including the risk of invasive breast cancer, became too great. Even the relatively short-term use of estrogen plus progestins significantly increases the risk of breast cancer. Using unopposed estrogen and estrogen plus progestin for long periods of time has also been associated with increased ovarian cancer risk.
In contrast, observational studies suggest that in menopausal women, the use of estrogen with progesterone may be associated with a lower risk of breast cancer when compared to the use of estrogen and synthetic progestin (PMID: 27456847).
In fact, women with a progesterone deficiency have 5.4 times the risk of premenopausal breast cancer when compared to women with normal hormone levels. Progesterone is highly protective of breast cancer (PMID: 7304556).
For women on hormones it can be helpful to:
Look for “micronized progesterone.” This is bioidentical and often derived from wild yam. Products that list “norethindrone,” “medroxyprogesterone acetate (MPA),” etc. are synthetic progestins.
Don’t be afraid to clarify with your doctor: “Is this bioidentical progesterone or a synthetic progestin?” Many providers still use the terms interchangeably.
Look for USP Progesterone: If using a cream, the ingredient should say “USP progesterone.” That’s the standard for bioidentical formulations.
While, progestins are commonly used in birth control, fertility treatments, and menopause therapies, for my patients, I like to use real, bioidentical progesterone, especially when estrogen is part of the therapy.
The word progesterone should not be used to describe progestins, yet this confusion still shows up in medical literature and mainstream reporting. Getting the terminology right is so important because it has real health consequences.
3. “If a cancer is progesterone-receptor positive, progesterone contributes to its growth.”
Progesterone receptor–positive (PR+) breast cancer refers to tumors that express receptors for progesterone. These receptors act like molecular switches. When the hormone binds, it can influence the cell’s behavior, including metabolism, growth, and gene expression.
PR+ tumors are almost always also estrogen receptor–positive (ER+), because estrogen signaling triggers the production of progesterone receptors. Together, ER and PR status help define a broader group called hormone receptor–positive (HR+) breast cancers, which account for about 75% of all breast cancer cases.
Historically, it has been assumed that because estrogen receptors (ER) promote tumor growth, progesterone receptors (PR) must do the same. But this is a misconception. PR is actually regulated by estrogen, meaning its presence often reflects an active estrogen signaling, not that progesterone itself is driving growth.
The role of progesterone itself has remained poorly understood, and this confusion has led to longstanding caution around its use in PR+ cancers. Although progesterone receptor–positive (PR+) tumors contain receptors for progesterone, this does not mean progesterone causes cancer:
The presence of a receptor simply means the cell can respond to the hormone, not that the hormone is harmful. Clinical treatment strategies have historically focused on blocking estrogen and have largely avoided giving progesterone, often out of caution rather than evidence.
Estrogen, not progesterone, has been repeatedly and conclusively shown to drive tumor growth in hormone receptor–positive breast cancers. Estrogen directly stimulates cell division in breast tissue, and decades of research, from molecular studies to large-scale clinical trials, proved that blocking estrogen with therapies like tamoxifen or aromatase inhibitors reduces recurrence and improves survival.
Because progesterone does not cause cancer, there are no widely used drugs specifically designed to block progesterone in cancer treatment. Antiprogestins like mifepristone exist, but they are not routinely used in clinical practice because they have not shown consistent benefit or improved outcomes.
The assumption that progesterone might also cause cancer growth arose largely because PR+ tumors express the receptor, not because there was strong evidence showing that progesterone itself promotes malignancy. In fact, there is no evidence that natural progesterone independently causes tumor growth.
We know mechanistically that progesterone has several anticancer effects. Progesterone:
Induces cellular differentiation. It encourages cancer cells to mature into more specialized, less aggressive forms.
Inhibits cell proliferation. Progesterone slows down the rate at which cancer cells divide, partly by suppressing growth-promoting genes like cyclin D1 and MYC.
Reprograms estrogen receptor activity. It alters how estrogen receptors bind to DNA, reducing estrogen’s tumor-promoting signals in ER+/PR+ cancers.
Promotes apoptosis. It triggers programmed cell death by upregulating p53 and downregulating survival genes like BCL-2.
Improves mitochondrial function. It supports cellular energy production and reduces oxidative stress by enhancing mitochondrial efficiency and promoting a shift away from glycolysis toward oxidative phosphorylation.
Reduces inflammatory signaling. It suppresses inflammatory mediators like IL-6, COX-2, and TNF-α, which contribute to tumor growth and immune evasion
Stabilizes the tumor microenvironment. It helps maintain tissue integrity and reduce oxidative stress, creating a less favorable environment for cancer growth.
There is no known mechanism by which progesterone promotes cancer. Unlike estrogen, which directly encourages proliferation, progesterone’s main role is to support differentiation and stabilize tissue structure.
This is why focusing only on receptor status can be misleading. Just because a tumor expresses progesterone receptors does not mean progesterone is promoting its growth. What matters is the biological action triggered by the hormone, and in the case of progesterone, that action tends to limit rather than encourage uncontrolled division.
Because of its protective effects, progesterone may actually be beneficial for PR+ cancers.
In a 2015 study published in Nature, researchers demonstrated that activating the progesterone receptor (PR) with natural progesterone could actually reprogram estrogen receptor (ER) activity in ER+/PR+ breast tumors, making them less aggressive and more responsive to treatment. Using natural progesterone on PR+ positive tumors reduced cell proliferation and promoted differentiation, effectively softening the tumor's behavior. These effects were even more pronounced when progesterone was combined with conventional anti-estrogen therapies like tamoxifen or aromatase inhibitors. This combination produced a greater anti-tumor effect than either treatment alone.
In another study published in the Annals of Clinical & Laboratory Science, researchers conducted in vitro experiments using human breast cancer cell lines to investigate the direct effects of natural progesterone on tumor behavior. They specifically focused on T47-D cells, which express the progesterone receptor (PR), and compared them to PR-negative cancer cells. Interestingly, they found that natural progesterone significantly inhibited cell proliferation, by up to 90%, and triggered apoptosis (programmed cell death) in the PR-positive cells. Authors concluded that natural progesterone not only slows cancer cell growth but actively helps eliminate malignant cells.
In a 2017 study published in Cellular Oncology, researchers investigated the effects of natural progesterone on breast cancer cell behavior. They conducted in vitro experiments using both progesterone receptor-positive (PR+) and PR-negative (PR−) human breast cancer cell lines. The study found that progesterone treatment led to a significant reduction in cell invasion and migration in both PR+ and PR− breast cancer cells. This inhibitory effect was associated with reduced the activity of certain proteins (like FAK and cofilin) that normally help cancer cells move and invade nearby tissue. The findings suggest that natural progesterone can suppress the invasive behavior of breast cancer cells through mechanisms independent of progesterone receptor expression.

Natural progesterone should be recognized as a valuable tool in treating hormone-sensitive breast cancers, even in PR+ tumors, where the receptors it needs are still present and active. “Physicians should have no hesitation prescribing natural progesterone” (Lieberman and Curtis, 2017).
4. “Men don’t need progesterone.”
Another common misconception is that progesterone is only relevant to women. Because it is associated with the menstrual cycle and pregnancy, it is often assumed to have little or no importance in male physiology. However, progesterone is important for men too.
Men produce it primarily in the adrenal glands and testes, though levels are naturally lower than in women. Even so, progesterone still participates in maintaining hormonal balance. When progesterone levels are insufficient relative to estrogen, men can experience symptoms associated with estrogen excess, including fatigue, mood changes, decreased libido, and metabolic disturbances.
Progesterone plays a role in male fertility itself. Studies have shown that when sperm encounter progesterone in the female reproductive tract, it helps activate the processes that allow sperm to successfully fertilize an egg (PMID: 10928417)
Research has shown that progesterone can support prostate health by opposing the growth-promoting effects of estrogen in prostate tissue.
Progesterone also plays an important role in the male brain. It functions as a neurosteroid that helps regulate mood, stress, and cognitive stability. Progesterone and its metabolites interact with neurotransmitter systems involved in calming neural activity. Low levels have been associated with increased anxiety, irritability, and stress sensitivity.
Because progesterone is naturally lower in men, therapeutic dosing dosing must be lower as well: “After the age of 35 or 40, many men experience a decline in their resistance to stress, corresponding to the decline in protective substances such as DHEA and progesterone. Often a small amount of progesterone, 5 or 6 mg…can make a difference, sometimes lasting for a few days or more. Paying close attention, the effects are usually noticeable within about half an hour. At a certain level, progesterone can antagonize the effects of testosterone; in younger men. That effect passes within a day of stopping the progesterone" (Dr. Ray Peat, 2021).
Calling hormones “male” or “female” hormones can have real consequences because it shapes how they are used—or ignored—in medical treatment. In reality, the endocrine system does not follow such simple categories. Men produce progesterone and estradiol, and women produce testosterone. Each hormone participates in a broader network that supports metabolism, brain function, and reproductive physiology in both sexes.
The truth is, most doctors aren’t trained to use progesterone therapeutically. In conventional medicine, hormones are often viewed only through the lens of stimulation and suppression. Estrogen is a driver of growth, and progesterone is often assumed to act the same way. But that’s not how it works biologically.
Unfortunately, because natural progesterone can’t be patented, there’s been little incentive to study it. What we do have are decades of independent research showing that progesterone receptor–positive cancers often have better outcomes, that progesterone supports healthy cell differentiation, and that it may help treat cancer and countless other conditions (PCOS, endometriosis, autoimmune conditions, etc).
If you or someone you love is navigating hormone imbalances or cancer, it’s important to become your own advocate: read, research, and understand your options. Progesterone isn’t the enemy. And I believe the more we understand it, the more we can empower both women and men to approach cancer treatment with both science and hope.
